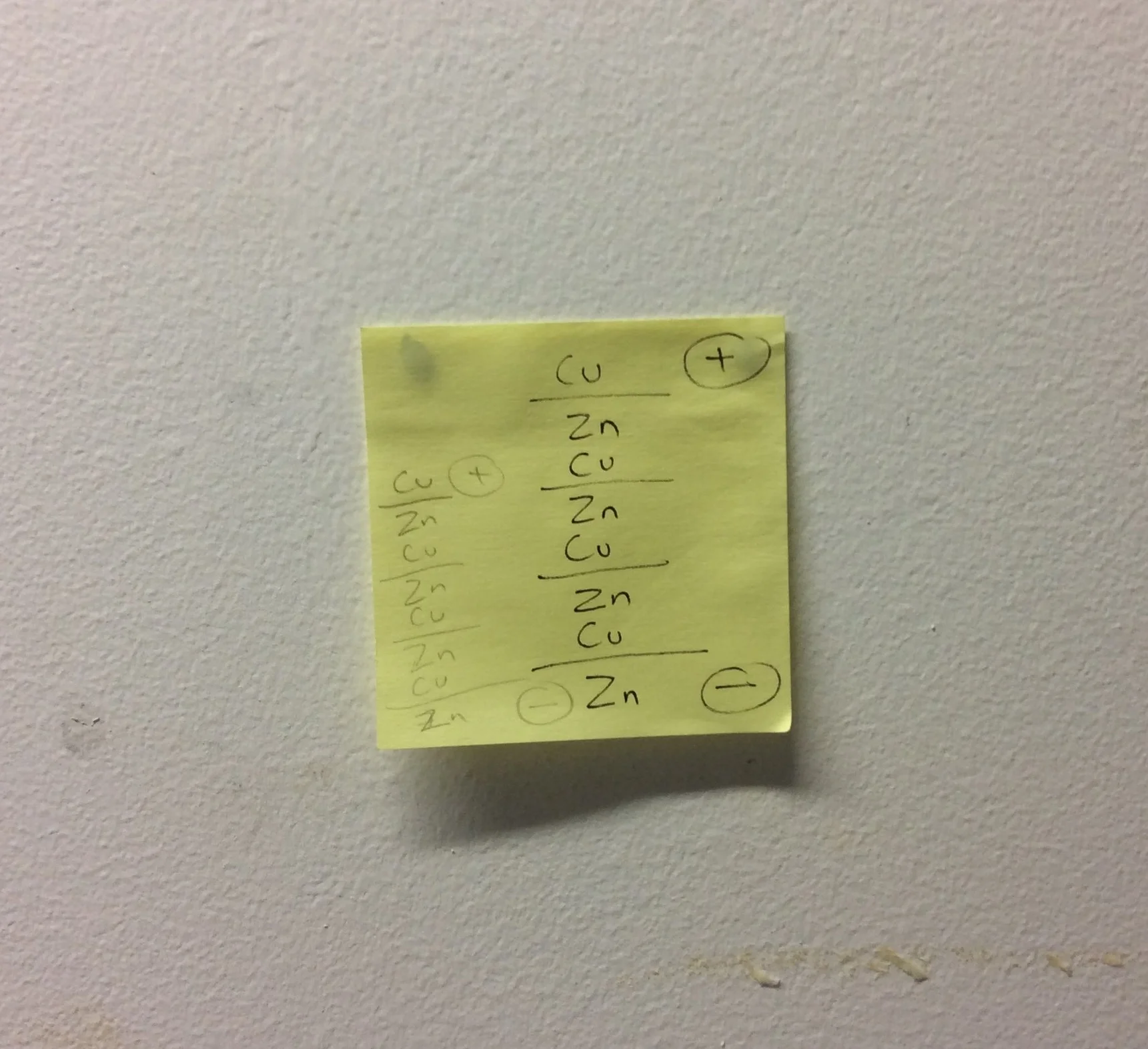Batteries (When the Power Runs Out)
Electricity begets metal begets electricity.
Discovered by Alessandro Volta in 1799, the voltaic pile was the first electrical battery that could continuously provide an electrical current. His original design consisted of sequentially ordered zinc and copper blanks, interspersed with an electrolytic-soaked fabric. This discovery enabled a rapid series of scientific discoveries in the 19th century, including the electrical decomposition (electrolysis) of water into oxygen and hydrogen, and the isolation of chemical elements sodium, potassium, calcium, boron, barium, strontium, and magnesium.
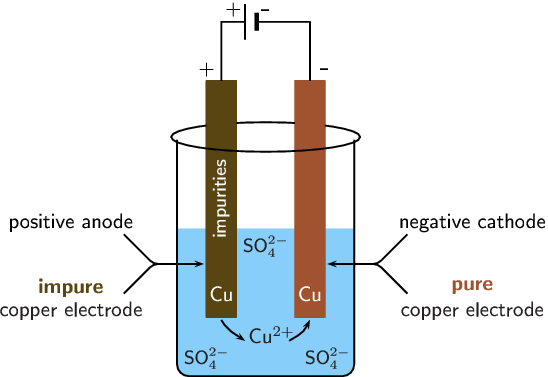
Electrochemical processes (that is, chemical reactions catalysed by electrical current) are presently used widely in heavy industry, most notably in refining pure metals from their earth encrusted ores. Electrorefining is a common process used in purifying electronics-grade copper.
Process
I wished to create a simple artwork / experiment that honoured the starting point of all electrical circuits by remaking Volta's beautifully simple zinc and copper voltaic pile.
However, making these batteries lead me down another path.
Returning to my studio, I found the week old remnants of my first voltaic pile to be surprisingly beautiful. Freshly disassembled, the corroded copper and the zinc plates were eerily reminiscent of stark landscapes, the copper in particular bringing to mind images of Simryn Gill's Eyes and Storms, aerial photographs of Australian open pit mines, dams, and other large scale human disruptions of the land. While initially I'd been wanting to create a power source, I became captivated by the simple expression of spent electrochemical energy evident in these small metal blanks.
With the spent cells now fully exposed to the atmosphere they continued to deteriorate. Over weeks and months the zinc washers became a rusty red-brown, and the copper caked over with white saline residue, the colours morphed, and eventually dulled. Over the following months I continued making voltaic cells, experimenting with different electrolytic solutions (usually a combination of sodium carbonate and sea salt, but i did attempt to melt down a few crystal speakers and use this solution) and plate sizes in order to get the most voltage out of my homemade cells. I also trialled various methods of cleaning the spent metal to impede the corrosion process. I found that methylated spirits and varnish helped to slow the oxidisation process, although nothing was able to stop the corrosion outright.
End result: Exhibition
Three variations on the voltaic cell idea was presented in the exhibition Source & Return at Firstdraft Gallery in mid 2017 with the title Batteries (when the power runs out).
In the first work, many small (approx 30x30mm, and 50x50mm) pieces of zinc, copper, cloth, and salt were made into batteries and the spent cells collected and presented in grids and piles. Innumerable patterns and colours resulted, with each piece continuing to oxidize over the weeks and months. The piles where exhibited alongside glass jars of methylated spirits stained with rust, another byproduct of this process -- used to slow the corrosion process of the metal. On the other side of the methylated spirit jars sat piles of fresh battery ingredients: copper, zinc, cloth, and salt.
In the second work, large plates (300x300mm) of zinc and copper were stacked in between with salt-water soaked cloth, creating a giant voltaic pile. This pile powered two bright LEDs for approximately 20 hours, before the electrochemical process no longer produced enough current. Once spent, the large metal plates where floor mounted on raw timber rails, and the salt-stained cloth was hung on the wall with copper nails. Again, the patterns and colour produced by the electrochemical process are unique and unstable.
Lastly, a small working voltaic pile was installed in the gallery space, complete with a bright white LED tucked within a rolled up copper-shim lamp shade. Affectionately referred to as the Salt Lamp, the handmade battery was replaced every few days of the exhibition, the spent remnants then added to the slowly corroding piles of metal and cloth.